GENERICS (UK) LTD V DAIICHI PHARMACEUTICAL CO LTD – ENANTIOMER OF A KNOWN COMPOUND HELD PATENTABLE BY UK COURT OF APPEAL
By Paul Cole
The present dispute concerned the patentability of the enantiomeric quinoline antibiotic levofloxacin covered by Daiichi’s expired EP (UK) 0206283 and by a supplementary protection certificate (SPC) based on it.
The racemic compound ofloxacin was known, and the unchallenged background situation as regards resolution was as stated at first instance by Kitchin J.at para. [110]:
“Overall, I think a relatively clear picture emerges. The quinolone field was unusual in that workers recognised the need for and perceived an opportunity to discover new chemical entities of ever greater efficacy. The discovery of norfloxacin, ofloxacin and ciprofloxacin put great pressure on researchers to identify new quinolones having even better bacterial profiles. It was here their energies were primarily directed. Hundreds of chemists were making new compounds each year and, not surprisingly, they would opt not to make a compound if its synthesis was difficult. Compounds which were difficult to make would get a low priority in the laboratory. This was not an environment conducive to the investigation of stereochemistry. Resolution of a racemate might result in a twofold increase in activity, at best, and it could be a good deal worse. It was quite possible that activity might lie more in one enantiomer than the other, but not greatly so. Moreover, resolution might prove very difficult to achieve. As a result, for many chemists, including those at Sterling-Winthrop, I am satisfied that resolution of racemates was something that would simply not have occurred to them at all. For others, it plainly did. But it was not a routine path to follow and, for the ordinary chemist, I believe it was something which he might well not have considered and, if he did, then it would not have been a high priority, absent some particular reason for doing so.”
The background situation as regards ofloxacin was quite different, according to Generics. A related tricyclic quinoline antibiotic called flumequine (I) had been disclosed.
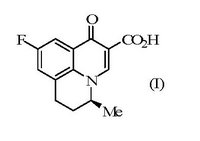
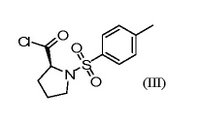
Racemic flumequine could not be resolved. But it had been disclosed that the enantiomers had been made and tested, and that an enantiomer (I) had potent antibacterial activity whereas the other enantiomer was only weakly active. How resolution had been achieved was disclosed in a poster shown for about two hours at a symposium in Toronto and involved use of a stereospecific intermediate (II). A weakness in the Generics case was that it had been resolved only using the N-tosyl-L-prolyl chloride (III) which was a non-standard resolving agent.
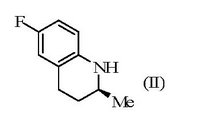
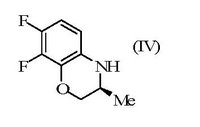
Generics averred that a skilled person would have been motivated to investigate whether the same marked difference in activity was demonstrated by the enantiomers of ofloxacin, and that an obvious route to those enantiomers was available starting from intermediate (IV) which was structurally similar to intermediate (II), would also have been expected to be resolved by the resolving agent (III) and would have permitted levofloxacin (V) to be obtained.
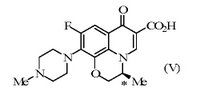
The Court of Appeal rejected this argument. Use of an unusual resolving agent suggested difficulty and unpredictability, otherwise a standard resolving agent would have been used, and the absence of a track record for the resolving agent made it less apparent that it might be generally useful. It was relevant that the generally accepted method of resolution started from the final product, not from an intermediate. Kitchin J. had correctly found that the structure of the intermediate (IV) was not part of the common general knowledge. Even if the unimaginative skilled person had found the intermediate (IV) and had recognised its similarity in structure to intermediate (II), “having the wit to draw conclusions from that” would have involved a level of invention. Furthermore, the synthetic reaction might or might not have worked and the skilled person had other things to try. In coming to the conclusion that the proposed route was not attractive enough, Kitchin J. had made no error of principle and had properly carried out the balancing task of forming an overall value judgment, which is so often the task of the first instance judge. In his concluding remarks concerning inventive step, Sir Robin Jacob said:
“I am not sorry to reach this conclusion. Daiichi’s work led to a better medicine than ofloxacin. Levofloxacin is not just twice as active as ofloxacin (which might have been expected) but is a lot more soluble and less toxic than was predictable. It can be used in higher dosages than might have been expected with corresponding medical benefit. Only a curmudgeon would say there was no invention here.”
The Court further held that the SPC had been validly granted. Earlier marketing authorisations did not give Daiichi the right to market levofloxacin as such, and it had to get marketing authorisation on the basis that it was a new drug. The research that lead to levofloxacin gave what was for all practical purposes a new drug. It was not realistic to regard ofloxacin as no more than levofloxacin with an impurity. Policy is aimed at preventing successive SPCs for mere minor variants of an active substance, which was not the case here. Levofloxacin was a novel and inventive improvement over ofloxacin, not a minor variant and had its own distinct activity, bioavailability and toxicity.
Comment
The Windsurfing test in the UK resembles the Graham v John Deere test in the US insofar as both provide structured preparation to put the court into a better position for reaching its decision, but when that preparation is complete leaves it with the task of reaching a decision according to the evidence. Under these tests there can be no bright line rule or standard: each case must be decided according to the evidence. Claims to individual enantiomers fall to be evaluated under these tests in the same way as claims to other new chemical entities and an enantiomer may, or may not, be inventive depending on the factual background of the individual case. However, in a decision earlier this year, the House of Lords upheld the patentability of the (+) epimer of citalopram, see Generics (UK) Limited v H Lundbeck A/S [2009] UKHL 12, and it will be apparent that the UK courts are likely to uphold claims to epimers where these can be shown to be the result of more than routine research.
It is black letter law that a route is obvious to try if there is a reasonable expectation of success, and the level of
expectation needed depends on the facts of each case. The Court approved and applied an observation of Kitchin J. in Generics (UK) Ltd v H Lundbeck A/S [2007] RPC 32 , para 72:
“The question of obviousness must be considered on the facts of each case. The court must consider the weight to be attached to any particular factor in the light of all the relevant circumstances. These may include such matters as the motive to find a solution to the problem the patent addresses, the number and extent of the possible avenues of research, the effort involved in pursuing them and the expectation of success.”
The Court recognised that there may be a number of obvious routes, and that there is no rule of law that requires only that option that is likely to be tried first or second to be treated as obvious, see Brugger v. Medic-Aid [1996] R.P.C. 635 at page 661 line 6 and Palmaz’s Patents [2000] RPC 631. However it held that this did not mean that a skilled person will pursue every avenue relentlessly when there are only the mildest of motives for doing so. In evaluating reasonable expectation of success it is now important to consider without hindsight the attractiveness of the necessary line of research at the time and in all the circumstances where the invention was made.
Paul Cole
Chartered Patent Attorney
CIPA authorised IP litigator
Generics v. Daiichi Pharmaceutical
Back to All Resources